In the race to bring new therapies to market in 2026, pharmaceutical R&D translation for drug discovery documents has become a make-or-break factor. Research teams generating terabytes of molecular data, assay results, and preclinical reports need every detail rendered with absolute precision across languages—because one misplaced term in a kinase inhibition profile or a binding affinity table can cascade into regulatory questions, trial delays, or outright rejection. For companies targeting FDA or EMA submissions, these translations aren’t optional extras; they’re the bridge that turns laboratory breakthroughs into approved medicines.
The numbers tell a stark story. The life sciences translation services sector is projected to reach USD 1.84 billion in 2026, growing at a compound annual rate above 8 percent through the early 2030s. At the same time, global pharmaceutical R&D spending continues its upward trajectory, with biopharma companies investing hundreds of billions annually to sustain innovation pipelines. Yet the average cost to develop a single new drug now exceeds $2 billion, and every month of delay in documentation review can erase tens of millions in potential revenue.
Why a Single Translation Slip Can Derail Years of Discovery Work
Consider the molecular level, where language must be as exact as the chemistry itself. Terms like “allosteric modulator selectivity,” “CRISPR-Cas9 off-target editing efficiency,” or “pharmacodynamic biomarker correlation” carry decades of scientific consensus. Translate them loosely and the meaning shifts. A European partner reading a Japanese-origin study on a novel oncology target might interpret “moderate affinity” as “high potency” if the nuance is lost—leading to mismatched expectations in joint development or flawed dose-escalation plans in Phase I trials.
Real-world consequences are brutal. Regulatory bodies demand consistency across the entire Common Technical Document (CTD). The FDA requires full English equivalents for any foreign-language source material in NDAs or BLAs, while the EMA conducts linguistic reviews of product information in all official EU languages before granting marketing authorization. A deviation flagged during these checks doesn’t just slow the clock; it can trigger complete response letters, additional studies, or even clinical holds. Industry analyses show that documentation-related delays routinely add hundreds of thousands to millions per day in lost opportunity costs.
Molecular Terminology in Practice: Where Precision Matters Most
Take a recent example from a mid-sized biotech’s antibody-drug conjugate program. The source report, drafted in German, described a linker chemistry with the phrase “cleavable unter lysosomalen Bedingungen.” A non-specialist translator rendered it simply as “cleavable under lysosomal conditions.” Technically correct—but missing the critical qualifier that the cleavage rate had been measured at pH 5.0 with a specific cathepsin B enzyme concentration. Without that detail, the U.S. regulatory reviewer questioned the stability data. The sponsor had to rerun stability assays and resubmit, pushing the IND filing back by nine weeks and burning through an extra $1.2 million in bridging studies.
Or look at gene therapy vectors. The term “self-complementary AAV” (scAAV) versus “single-stranded AAV” (ssAAV) isn’t interchangeable. One carries double the genetic payload and different immunogenicity risks. In a 2025 project involving a rare-disease candidate, inconsistent rendering across an Italian preclinical report and its English translation led to mismatched vector genome copy-number calculations in the CMC section. The FDA requested clarification, extending the review cycle.
These aren’t isolated anecdotes. They reflect a pattern: molecular biology evolves faster than general language models can keep up. Subtle distinctions in stereochemistry, polymorphism nomenclature, or pharmacogenomic haplotypes require translators who have spent years reading the primary literature, not just dictionaries.
The Edge That Only a Pharmaceutical PhD Team Delivers
Generalist agencies might promise speed and cost savings. What they cannot guarantee is the scientific intuition that catches an error before it reaches the regulator. That’s why leading sponsors now insist on dedicated pharmaceutical PhD teams—experts who hold doctorates in pharmacology, medicinal chemistry, or molecular biology and who translate full-time.
These specialists don’t just convert words; they validate scientific intent. They cross-check against ICH guidelines, EMA reflection papers on quality, and FDA’s latest Q&A documents on CMC documentation. They maintain terminology glossaries that evolve with every new modality—whether it’s mRNA, bispecific antibodies, or proteolysis-targeting chimeras (PROTACs). The result is a submission package that reads as if it originated in the target language, eliminating the “translated feel” that often triggers extra scrutiny.
Data-Backed Confidence in 2026 Pipelines
Global medicine use is forecast to keep climbing through 2030, with defined daily doses approaching four trillion annually. That growth rests on the steady output of novel active substances—381 launched worldwide between 2020 and 2024 alone, according to IQVIA tracking.
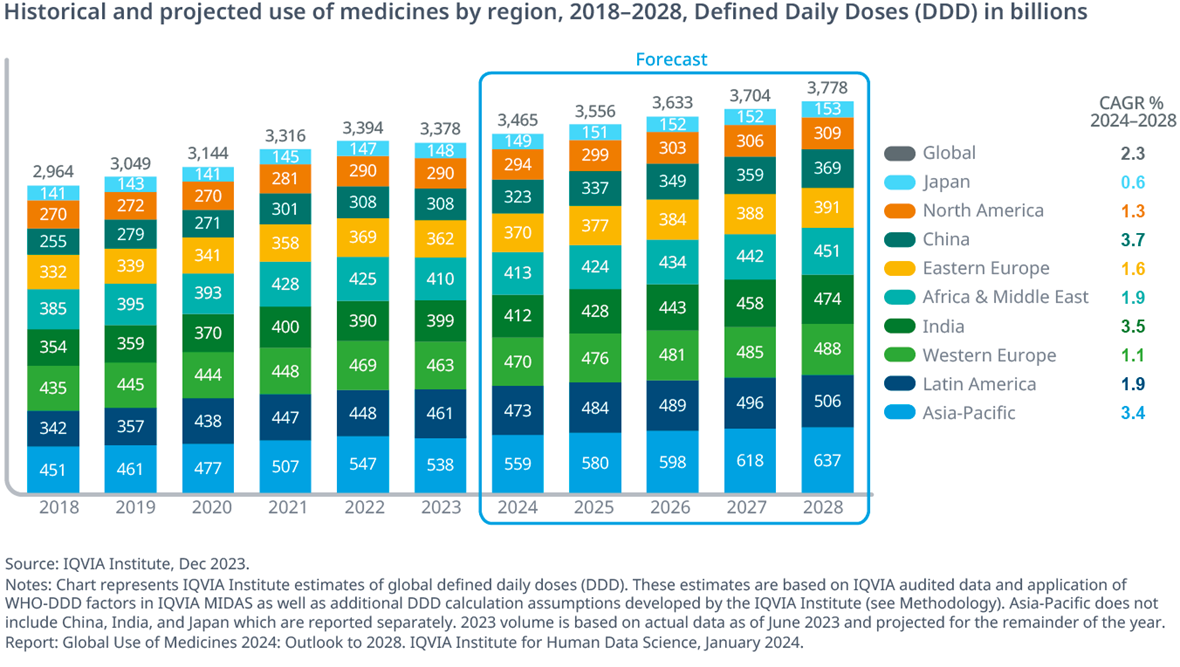
These charts underscore the pressure: more compounds, more complex science, more languages involved in global trials and manufacturing. The translation layer must scale without sacrificing accuracy.
Choosing the Right Partner for Drug Discovery Documents
When timelines are tight and stakes are high, sponsors need more than a vendor—they need a strategic extension of their own R&D and regulatory teams. That means seamless integration with eCTD publishing systems, version-controlled glossaries, and audit-ready quality assurance processes that meet both ISO 17100 and client-specific SOPs.
For organizations that demand this level of reliability, Artlangs Translation offers exactly that expertise. Proficient in more than 230 languages and honed over years of focused work in translation services—including video localization, short drama subtitle localization, game localization, multi-language dubbing for short dramas and audiobooks, plus multi-language data annotation and transcription—their teams have delivered hundreds of successful cases that prove the difference domain depth makes. Whether your next milestone is an IND filing, a Phase III protocol across three continents, or a full NDA dossier, they turn complex R&D materials into assets that regulators trust and global partners understand—helping you move from discovery to approval without the hidden costs of rework or delay.











